
Visualização & relatórios
SAS Visual Analytics
Agora todos podem facilmente descobrir e compartilhar insights poderosos que inspiram ação.
Um único aplicativo para geração de relatórios, exploração de dados e analytics.
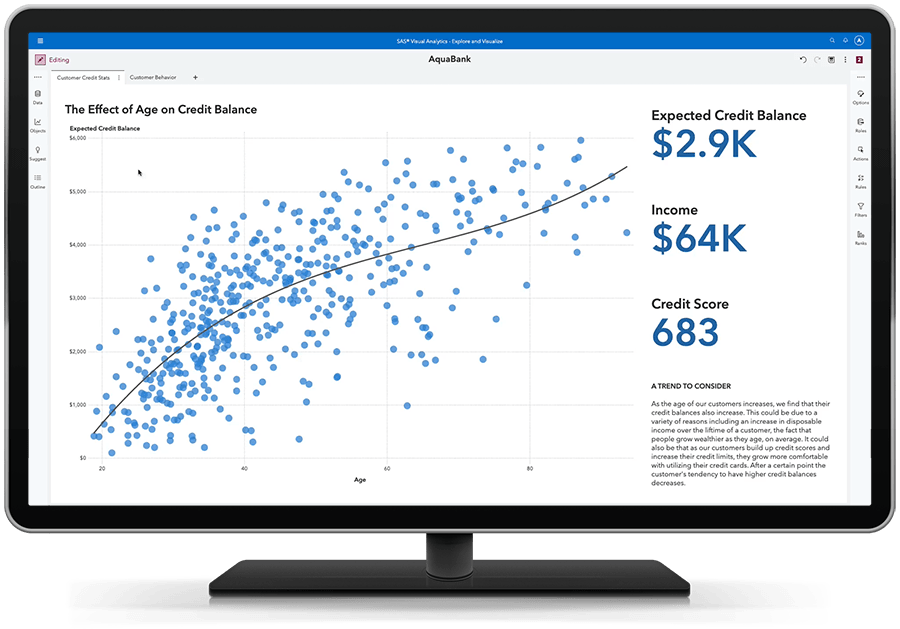
Veja o quadro geral - e as conexões subjacentes.
Localize rapidamente relações importantes em seus dados utilizando sugestões e medidas relacionadas claramente identificadas.
Use o machine learning e explicações de linguagem natural para encontrar, visualizar e narrar histórias e insights que sejam fáceis de entender e explicar. Descubra por que algo aconteceu, examine todas as opções e descubra oportunidades ocultas em seus dados. Destaque automaticamente os principais relacionamentos, outliers, clusters e muito mais para revelar insights vitais que inspiram a ação.
Transforme a visão em compreensão com recursos visuais dinâmicos.
Crie relatórios e painéis de controle interativos impressionantes. Resuma rapidamente as principais métricas de desempenho e compartilhe através da web e de dispositivos móveis. Os executivos e a equipe de linha de frente podem interagir e colaborar rapidamente com insights, dividi-los e cortá-los para encontrar suas próprias respostas e entender melhor o desempenho dos negócios.
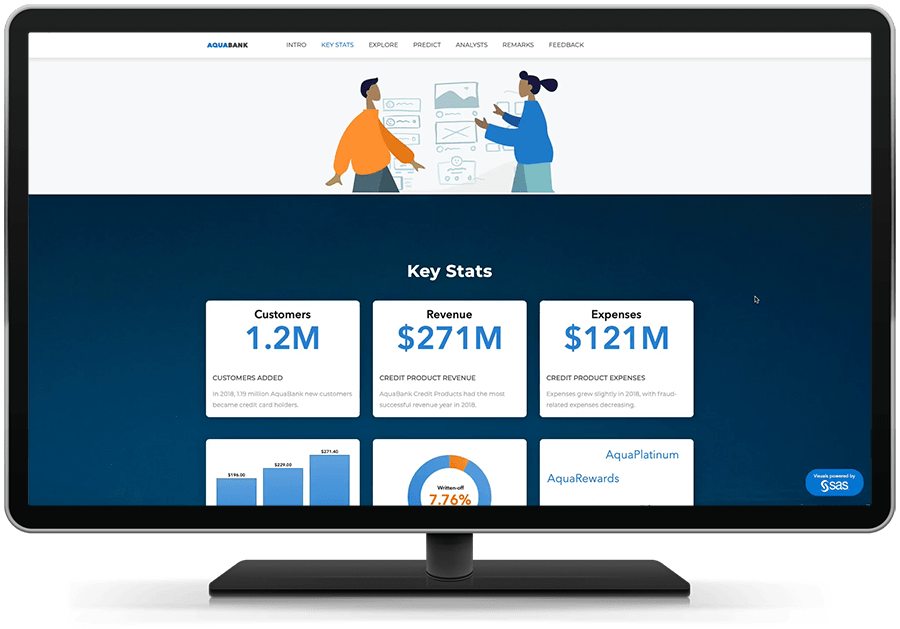
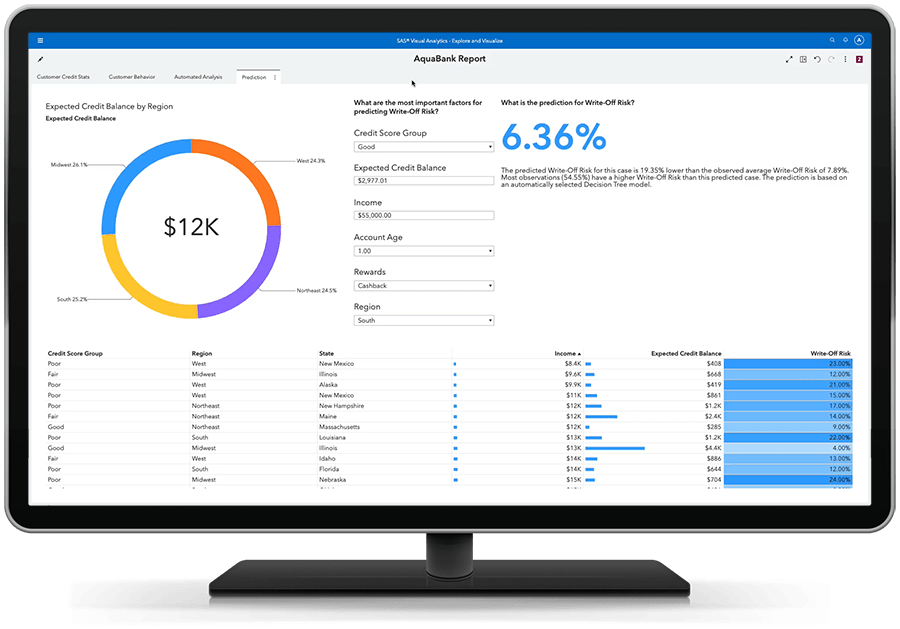
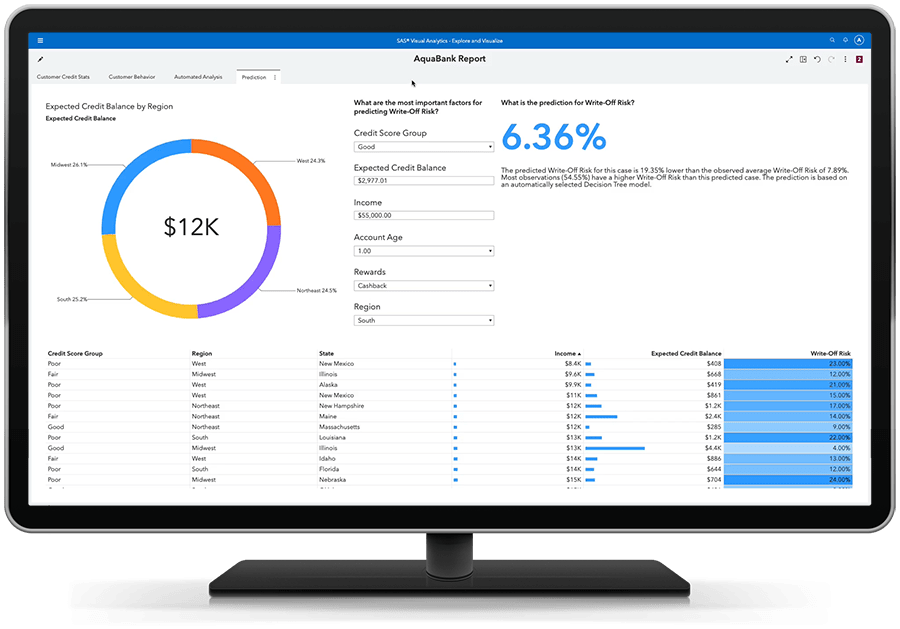
Obtenha respostas baseada por dados, resultados orientados por insights.
A análise preditiva fácil de usar permite que até mesmo os analistas de negócios avaliem possíveis resultados e tomem decisões mais inteligentes baseadas em dados – sem necessidade de programação. Algoritmos inteligentes reduzem a necessidade de experimentação manual. E você pode trabalhar em colaboração com especialistas para focar no que é mais relevante.
Adicione o "onde" ao "o quê".
Adicione contexto geográfico a suas análises e visualizações, combinando dados tradicionais com dados de localização. A análise de localização traz a dimensão “onde” para o primeiro plano, para que você possa analisar os dados de novas maneiras para obter uma visão completa antes de tomar decisões enquanto identifica oportunidades específicas de localização.
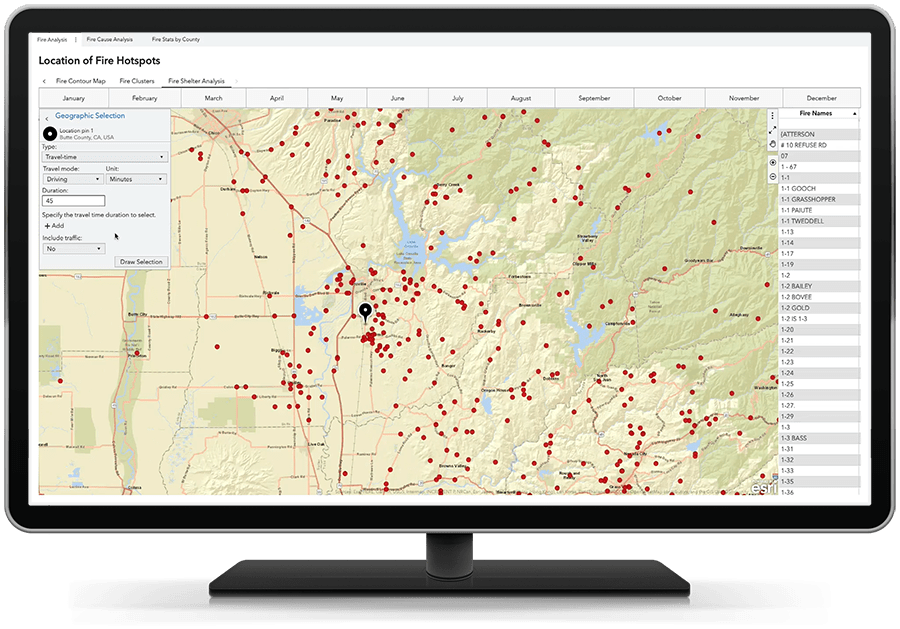
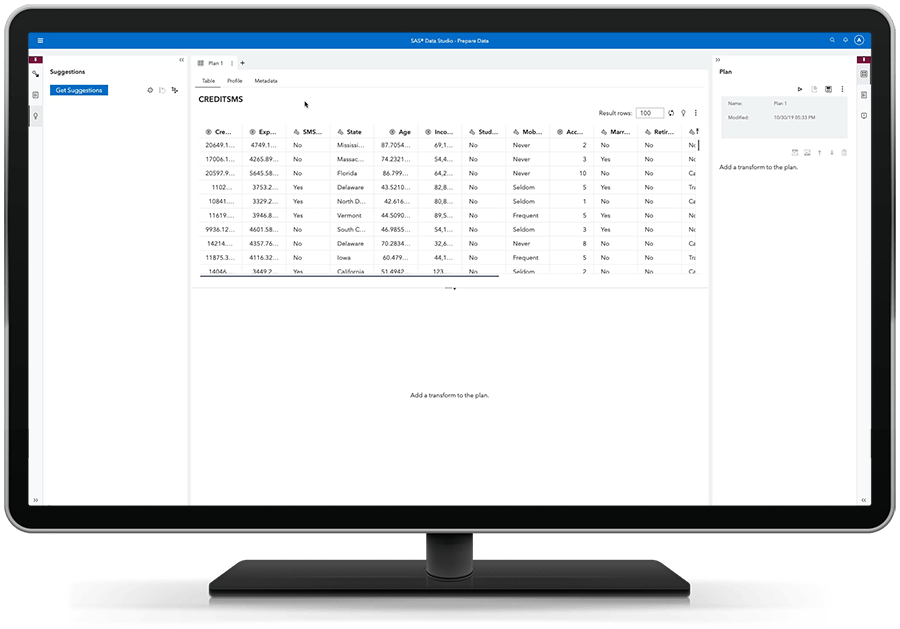
Agilize o processo de descoberta.
A preparação de dados de autoatendimento dá aos usuários comerciais a capacidade de importar seus próprios dados, unir tabelas, aplicar funções de qualidade de dados, criar colunas calculadas e mais - tudo com facilidade de drag-and-drop. Ao capacitar os usuários a acessar, combinar, limpar e preparar seus próprios dados de forma ágil - e confiável -, o SAS Visual Analytics facilita uma adoção mais rápida e ampla do analytics para toda a sua organização.
Obtenha insights habilitados por bate-papo.
Uma interface visual intuitiva e low-code permite a criação e implementação de chatbots personalizados e em linguagem natural. Obtenha respostas de texto, acesse dados, relatórios e visualizações e até mesmo aplique analytics e IA por meio de uma linguagem natural, interface de conversação. Configure bots dentro do ambiente SAS para facilitar o acesso a insights, ou conectá-los a serviços externos para implementá-los no mundo. Agora, obter insights a partir dos dados é tão fácil quanto enviar uma mensagem.
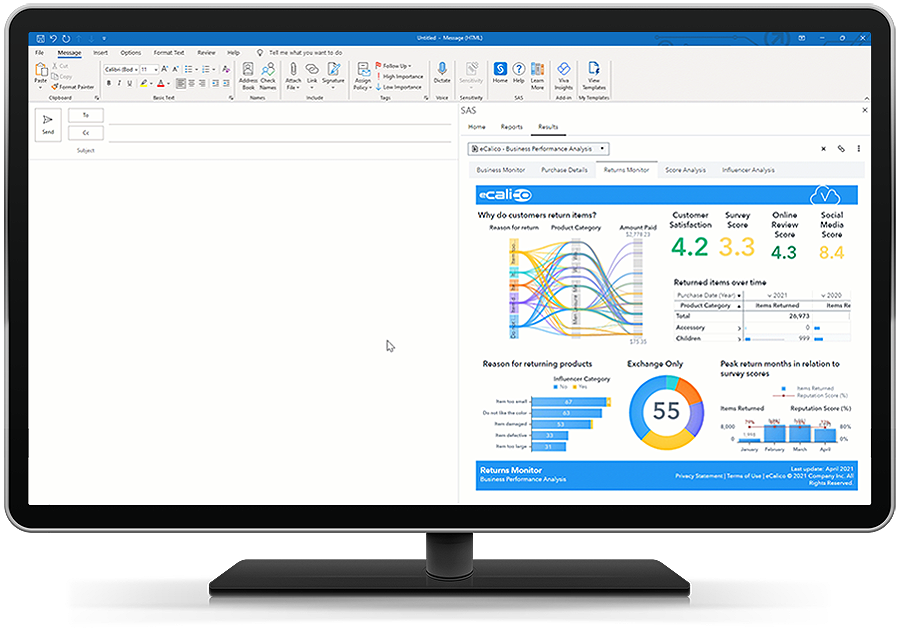
Aproveite o melhor de ambas as plataformas.
Use as ferramentas de produtividade da Microsoft do Microsoft 365 para acesso contínuo ao analytics do SAS. Aprofunde-se em seus dados, acesse e interaja com relatórios no SAS Visual Analytics e incorpore insights em seus aplicativos da Microsoft, como Excel e Outlook. Em seguida, compartilhe-os com seus colaboradores mais próximos.
Free Trial
Try the latest SAS Viya capabilities.
Get a free, 14-day trial of SAS Viya, which includes SAS Visual Analytics, as well as capabilities for the entire analytics life cycle.
Histórias de Sucesso de Clientes
Veja quem está trabalhando de forma mais inteligente com o SAS
SAS Visual Analytics ganha o crachá 'Users Love Us' do G2.

Principais recursos
Relatórios interativos. Descoberta visual. Analytics de autoatendimento. Escalabilidade e governança. Tudo a partir de um único e poderoso ambiente in-memory.
Acesso, preparação e qualidade de dados
Acesse, crie perfis, limpe e transforme dados usando uma interface intuitiva que fornece recursos de preparação de dados de autoatendimento com IA incorporada.
Criação de chatbot personalizados
Crie e implemente chatbots de linguagem natural personalizados por meio de uma interface visual intuitiva e de baixo código para insights habilitados para chatbot e experiências de conversação do usuário
Visualização de Dados
Explore dados visualmente, crie e compartilhe visualizações inteligentes e relatórios interativos por meio de uma única interface de autoatendimento. Análises aumentadas e capacidades avançadas aceleram os insights e ajudam a descobrir histórias escondidas em seus dados.
Painéis interativos, relatórios, BI e analytics
Vá diretamente de relatórios e exploração para análise e compartilhamento de informações por meio de diferentes canais, incluindo aplicativos do Microsoft Office, com uma única interface.
Analytics de autoatendimento
Previsão automatizada, busca de objetivos, análise de cenários, árvores de decisão e muito mais estão ao seu alcance, não importa qual seja seu nível de habilidade.
Análise de texto
Obter insights das mídias sociais e outros dados de texto e saber se o sentimento é positivo ou negativo.
Análise de localização
Combinar fontes de dados tradicionais (transacionais, clientes, operacionais, etc.) com dados de localização para análise em um contexto geográfico.
Análises aumentadas
Descubra as histórias reais escondidas em seus dados - em segundos. O software lhe dá automaticamente sugestões e identifica medidas relacionadas.
Insights incorporados
Incorpore insights visuais individuais dos relatórios do SAS Visual Analytics, construa histórias interativas ou incorpore todo o relatório ou página interativa em suas aplicações web personalizadas. Publique suas percepções que escalam com a flexibilidade de dados ao vivo ou snapshots de dados incorporados para contar histórias de dados.
SAS Visual Analytics Apps para dispositivos móveis
Use aplicativos móveis nativos para iOS e Android para visualizar e interagir com outras pessoas por meio de relatórios e painéis dinâmicos em tablets e smartphones.
Integração aberta para desenvolvedores
Adicionar visualização criativa com bibliotecas JavaScript de terceiros, como D3 e C3, com plena interatividade dentro do SAS Visual Analytics. Aproveite os recursos de desenvolvimento de código aberto para desenvolvedores e APIs REST para qualquer linguagem de cliente para acessar analytics, dados e serviços SAS.
Nativo da nuvem
A arquitetura do SAS Viya é compacta, nativa da nuvem e rápida. Se você preferir usar o SAS Cloud ou um fornecedor público ou privado de nuvens, você poderá aproveitar ao máximo seu investimento em nuvens.
Integração com Microsoft 365
A integração perfeita entre SAS para Microsoft 365 e SAS Viya permite que você incorpore insights analíticos poderosos de SAS dentro de suas aplicações Microsoft 365, estimulando a colaboração e conduzindo decisões mais bem informadas.
Suporte a idiomas
Os idiomas suportados incluem japonês, coreano, chinês simplificado, chinês tradicional, tailandês, alemão, espanhol, francês, italiano, polonês, russo, árabe, tcheco, dinamarquês, inglês, grego, croata, húngaro, hebraico, norueguês Bokmal, holandês, norueguês, português brasileiro, português, eslovaco, esloveno, sérvio, sueco e turco.
Demonstrações interativas
Confira estas demonstrações interativas do SAS Visual Analytics para ver como você pode explorar e entender dados de forma rápida e fácil para resolver uma variedade de questões industriais e comerciais.
- Serviços Bancários Banking & Risk InsightsGet a holistic view of risk and performance across regions, down to the individual counterparty level.
- Energia e Serviços Públicos Water Consumption & QualityMonitor the quality and consumption of this critical natural resource.
- Varejo Retail InsightsAnalyze regional store performance, and make data-driven decisions about retail business functions.
- Fabricação Warranty AnalysisAnalyze warranty claims to quickly identify potential issues and their underlying causes.
Explore mais sobre SAS Visual Analytics e além
Para navegar pelos recursos por tipo, selecione uma opção abaixo.
-
- Selecione o tipo de recurso
- Artigo
- E-BOOK
- Entrevista
- Estudo de caso
- História do cliente
- Infográfico
- PESQUISA
- POST BLOG
- RELATÓRIO DE ANÁLISE
- Série
- Trecho de livro
- Vídeo
- Webinar
- White Paper
- História do cliente Modernization initiative set to improve taxpayer compliance and moraleThe Malta Tax and Customs Administration automatically detects compliance discrepancies and filing errors with SAS Viya.
- História do cliente Jakarta Smart City uses IoT analytics to better serve residentsJakarta and SAS team up to create an award-winning approach to public services and disaster management.
- História do cliente Breaking the cycle of poverty through advanced analyticsCompassion International uses advanced analytics to help millions of children from impoverished nations secure food, medical care and educational resources to grow, learn and thrive.
- História do cliente Managing Dutch roads and waterways with intelligenceA modern AI, IoT and analytics platform powered by SAS Viya helps Rijkswaterstaat move from reactive to predictive infrastructure maintenance.
- História do cliente História do cliente Advanced analytics helps policymakers determine how new tax measures would affect citizensAscertaining the ‘winners’ and ‘losers’ of potential tax changes before regulations are implemented.
- História do cliente Optimizing cancer patient care with advanced analyticsSAS Viya helps create custom care pathways for Oscar Lambret Center cancer patients.
- História do cliente Automated safety reporting protects hospital patients in NorwayHelse Nord uses SAS to automate its hospitals’ processes and halves their workload.
- História do cliente Taking the guesswork out of production planningEuramax uses SAS to prevent production delays
- História do cliente Hospitals save time and money with real-time medical device tracking Jan Yperman Hospital uses Blyott and SAS Visual Analytics on SAS Viya to monitor hospital asset locations and ensure optimal purchasing decisions.
- História do cliente One airport operator is making every journey personal for passengersiGA Istanbul Airport creates personalized customer journeys using SAS® MarTech solutions
- História do cliente Improving data collection and modeling to accelerate predictive medicine effortsDompé farmaceutici uses SAS for predictive analytics and quantitative disease modeling.
- História do cliente Enhancing road safety with advanced analyticsBelgium’s FPS Justice uses insights from SAS Visual Analytics to crack down on excessive speeding and take more effective action against traffic violators.
- História do cliente Ensuring public transparency and one-stop access to crime data and analysisThe North Carolina Criminal Justice Analysis Center uses SAS Viya on SAS Cloud to improve data accessibility and efficiencies in gathering and reporting state and county criminal justice statistics to stakeholders, policymakers and citizens.
- História do cliente Making prisons and communities safer with data visualizationIowa Department of Corrections uses SAS Viya on SAS Cloud to better manage supervised populations, make informed decisions and improve public safety.
- Artigo Are you good at scoring?Credit scoring is the foundation for evaluating clients who apply for a loan (or other types of exposure for the bank). It is not unusual for it to take up to 12 months to build and deploy a new credit scoring model. Reforming the process will help minimize losses, increase earnings and reduce operational risk.
- História do cliente One of the largest multi-sport clubs in Portugal aims to win big with analyticsSport Lisboa e Benfica modernizes its operations with SAS Analytics, as it looks to expand internationally.
- História do cliente Norway’s largest telecommunications, media and technology provider adapts to rapidly changing customer expectations using a hybrid cloud approachTelenor relies on SAS Viya on Azure and SAS Customer Intelligence 360 to provide fast, personalized customer service based on real-time analytics.
- História do cliente Analytics helps major public health system run efficient programs and improve patient careThe Los Angeles County Department of Health Services relies on advanced analytics from SAS to meet federal regulations, ensure financial viability and better serve a diverse population of more than 10 million people.
- História do cliente It’s all in the research: Using AI to solve issues in health careWith the University of Alberta's new health data management and analysis platform, DARC, it can now increase research capacity and provide high-performance computing and data storage in a secure environment. SAS provided the university software to help make its platform thrive amidst a global pandemic.
- História do cliente Analytic models spotlight risky loansItaly’s Ministry of Economy and Finance uses advanced analytics on SAS Viya to quickly calculate risk on financial guarantees.
- História do cliente Italy’s second-largest hospital uses advanced analytics for effective pandemic responseGemelli University Hospital uses SAS solutions to predict admissions of patients in the intensive care units and impacts on the organization, from staffing hospital wards to effectively scheduling and managing COVID-19 vaccine administration.
- História do cliente Finland’s top retail bank applies AI to improve customer service and credit scoringS-Bank provides better customer service and faster, more accurate loan processing time using SAS Viya on Azure.
- História do cliente Real-time analytics helps telecom provider adapt to changing customer needs during global pandemic and beyondTelefónica Ecuador accelerates digital transformation, improves campaigns and achieves growth via intelligent decisioning powered by SAS.
- História do cliente Transforming the consumer banking experience through advanced analyticsCIMB Singapore uses SAS Viya to enhance business operations and keep pace with changing customer needs.
- História do cliente Delivery company relies on analytics to optimize logistics and swiftly meet changing marketplace dynamicsRelais Colis reinvents itself during global pandemic using SAS Visual Analytics to ensure continuity of business and quality of service.
- História do cliente Reaching 3.6 million football fans via comprehensive marketing analyticsThe Royal Dutch Football Federation relies on SAS Customer Intelligence 360 to better understand what motivates players and fans, engage audiences with personalized communications, and promote the love of the game.
- História do cliente Analytics helps ensure fair and balanced pay structures across Sweden’s public sectorTietoEVRY and SAS partner to help Sweden's largest employer organization, Adda, offer a nationally aggregated salary planning tool for municipalities and regions across the country.
- História do cliente Travel and tourism forecasts become more accurate with analyticsDER Touristik builds web-based planning tool with SAS to better predict future demand and quickly react to changes in the market.
- História do cliente A data-driven approach to whole person careRiverside County relies on data integration and analytics from SAS to improve the health and well-being of vulnerable Californians.
- Artigo From living on the streets to owning a profitable businessZoe Empowers, a ministry for vulnerable children living in life-threatening poverty in Africa and India, finally found a way to measure and reveal its incredible impact. Using SAS, they developed an "empowerment index" that measures improvements in housing, health, education and more for program participants.
- História do cliente Prison violence dramatically drops with analytics-based risk assessmentThe Indiana Department of Correction uses real-time data and predictive analytics from SAS to reduce violent attacks on staff by 50%.
- História do cliente Funding for ground-breaking childhood cancer research fueled by analyticsThe Kids’ Cancer Project uses SAS to improve fundraising efforts for vital cancer research.
- História do cliente A model of institutional research champions the value of analytics for allOklahoma State University uses SAS to create an analytics culture and increase student success.
- História do cliente Predictive analytics and AI deliver a winning fan experience The Orlando Magic uses mobile app data and machine learning to personalize marketing campaigns and analyze game data.
- História do cliente Forecasting accuracy brings ‘new energy’ to CameroonEnergy supplier Eneo balances supply and demand to boost efficiency, save millions and improve reliability with SAS Energy Forecasting.
- História do cliente Artificial intelligence and IoT analytics keep aircraft operational for crucial missionsLockheed Martin revolutionizes aircraft maintenance with the SAS Platform.
- História do cliente Property experts open analytics to new marketsNVM enhances real estate analytics with SAS Viya open architecture.
- História do cliente Boosting institutional research with data governance and self-service reportingThe University of Idaho invests in SAS to give administrators access to trusted, insight-rich data.
- História do cliente Next-day deliveries in hand with analyticsParcel delivery company Relais Colis adapts to online shopping revolution by using SAS Visual Analytics to automate reporting, shorten delivery times.
- História do cliente Automated laboratories improve uptime with analyticsPredictive service and maintenance keeps Siemens Healthineers lab tests running on time.
- White Paper Tar Heels Play to Win With SAS Analytics Learn how Carolina Athletics and its fundraising arm, The Rams Club, use SAS Analytics to deepen existing relationships and broaden a longstanding tradition of loyalty to all 28 of their university sports.
- História do cliente Smart data exploration advances K-12 public education programsThe South Carolina Department of Education depends on SAS to analyze data and properly fund and serve its school districts.
- Artigo Location intelligence: Adding geospatial context to BI Get tips for implementing a location intelligence data strategy from industry thought leader and data explorer, Jen Underwood.
- História do cliente Achieving academic and operational excellence through business intelligenceCurtin University uses SAS Visual Analytics to provide reporting across the organization.
- White Paper The Power of CollaborationThis white paper explains the need for and value of the collaboration and self-service capabilities that SAS Visual Analytics and SAS Office Analytics provide. It provides how-to specifics for incorporating SAS Visual Analytics results with Microsoft Outlook, Excel, PowerPoint, Word and SharePoint with plenty of screen shots, explanations and architecture diagrams for IT.
- Entrevista Location analytics: Why adding where makes BI betterLearn how location analytics can enrich your BI endeavors by blending geographic data, spatial analysis and business data for deeper insights.
- White Paper Redefine Your Analytics Journey With Interactive Data Exploration and Predictive AnalyticsWhat if your analytics journey were easier? It can be, in five easy steps. Learn how SAS Visual Analytics and SAS Visual Statistics together can provide a fast, fun way to explore data, build models and find the best performer.
- White Paper Data Visualization TechniquesA picture is worth a thousand words – especially when you are trying to find relationships and understand your data – which could include thousands or even millions of variables. This paper takes you from the basics of charts and graphs to visualizing big data using SAS Visual Analytics.
- E-BOOK Improving Customer ExperienceEveryone makes better decisions with easy access to powerful, interactive analytics – no matter the size of the business. This e-book profiles seven organizations that are using self-service data visualization and exploration to make big improvements in the way they work.
- White Paper Workforce AnalyticsThis paper explores how government HR functions can use advanced analytics, machine learning and AI to develop effective plans to meet hiring, retention and performance goals.
- White Paper TDWI Checklist Report: Gaining Business Value from Governed Analytics and Discoverydata governance
- História do cliente University uses analytics to ensure student successWestern Kentucky University uses data visualization and advanced analytics to make informed decisions.
- História do cliente Better reporting yields better understanding of risk managementSAS Visual Analytics helps Erste Bank Croatia tackle diverse data for accurate analysis.
- História do cliente Valencia College uses data-driven approach to improve outcomes in higher educationSAS Visual Analytics empowers administrators to easily manage enrollment, and adjust class availability and resources to help students succeed.
- White Paper The Use of Open Source is Growing. So Why Do Organizations Still Turn to SAS?This conclusions paper summarizes a session at the 2014 Hadoop Summit presented by Brian Garrett, Principal Solutions Architect at SAS Institute Inc. titled, “With the Rise of Open Source, Why Organizations Still Turn to SAS.” In it, he explains why corporate analytics requirements are best served when companies run both SAS enterprise-class analytics solutions and open source solutions together. Garrett explains how these technologies meet different needs and co-exist well. He also highlights recent SAS software enhancements that allow analysts to incorporate R algorithms into analytic processes as part of a comprehensive, enterprise-class SAS analytics platform.
- Artigo Is your data too big to visualize?Use these tips to find new ways to look at big data, so you can quickly collapse and condense the results in an intuitive fashion.
- Entrevista Data visualization: A wise investment in your big data futureData visualization technologies can help the practice of data-driven decision making really take hold. But putting data visualization software in the hands of business users? Is it crazy – or crazy smart?
- White Paper Fast and FuriousWhite paper for IT that explains the components and deployment options for SAS Visual Analytics.
- White Paper Building an Analytical Culture for SuccessAn ambitious, culture-centric project reshaped people’s attitudes about data and quickly returned more than a $1 million in cost savings. See the six guiding principles that led to success where three earlier attempts had failed.
- E-BOOK Augmented Analytics: The secret ingredient to better business intelligenceAugmented analytics breaks down the limitations of business intelligence (BI) and brings forward insights from data using AI and machine learning.
Ofertas relacionadas
Confira estes produtos e soluções relacionados ao SAS Visual Analytics.





